50 year old female with no reported/known medical history presents to the ED with hypoxia (SPO2 60%), respiratory distress, altered mental status, and hypotension.
Initial Vital Signs are: HR 120, RR 30, Blood Pressure 70/50, and SpO2 65% on room air with improvement to 85% on Non-rebreather mask.
She was immediately intubated, sepsis therapy initiated, and EKG obtained. Pt required significant FIO2 between 60-80% as well as Norepinephrine to maintain blood pressure.
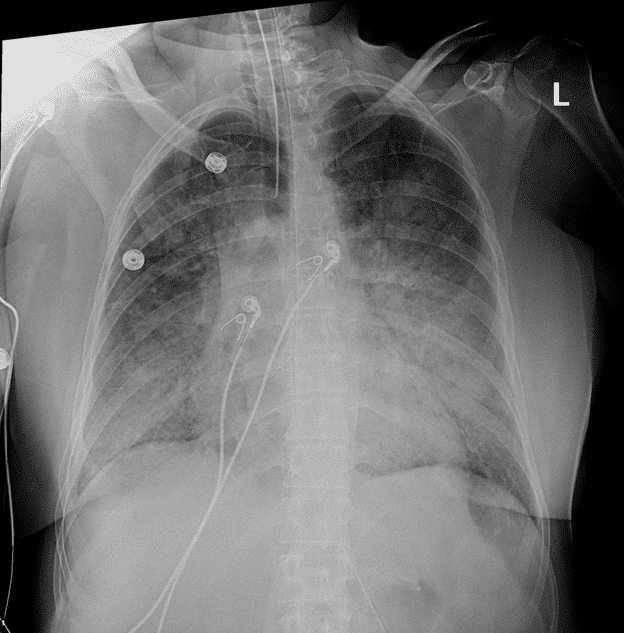
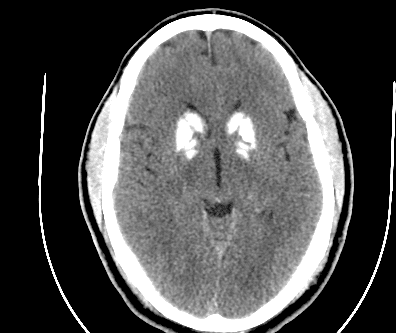
CT scan 1
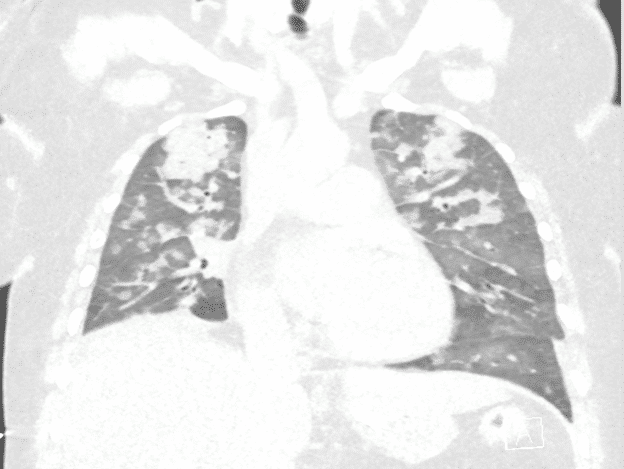
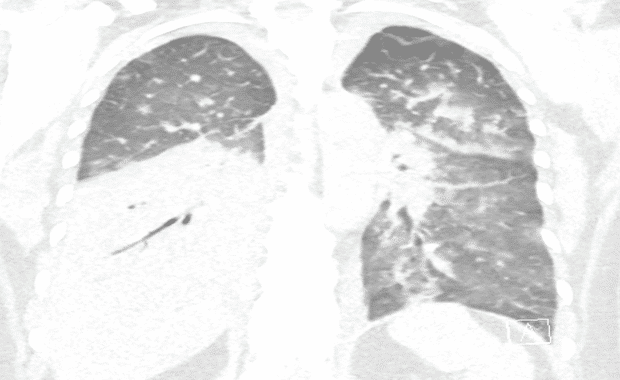
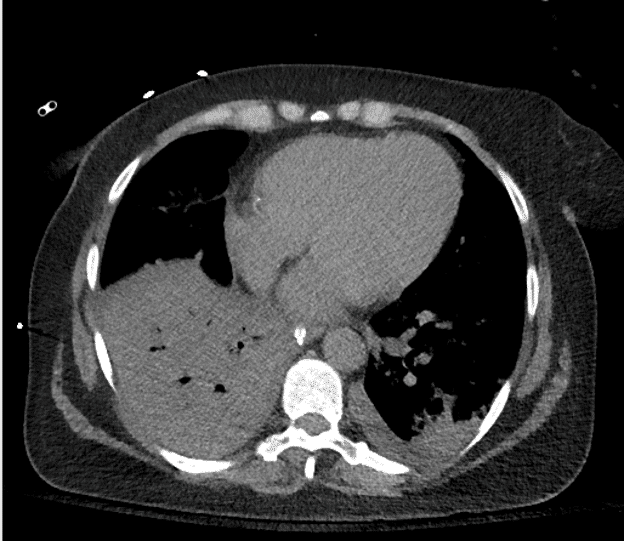
Initial Chest Xray and CT scans consistent with lobar pneumonia, however patchy infiltrates also noted and COVID-19 considered possible, therefore the patient’s isolation was continued from the ED to the ICU. Empiric antibiotics were started along with fluid bolus. Complicating matters, at the time, visitors were not allowed to come to the hospital and the patient’s family did not give EMS a good contact number so no collateral information was obtained for 3 days until the patient’s husband showed up at the hospital.
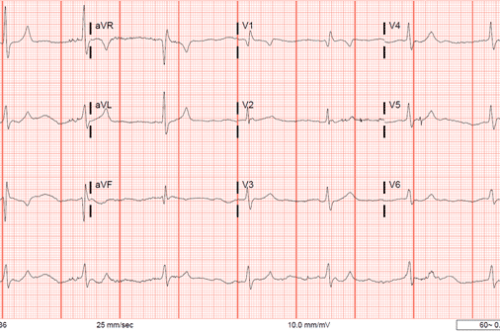
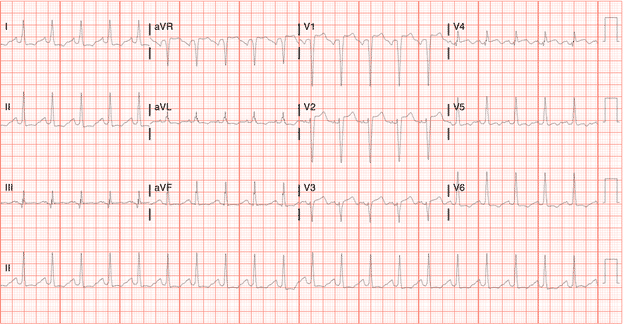
EKG 1
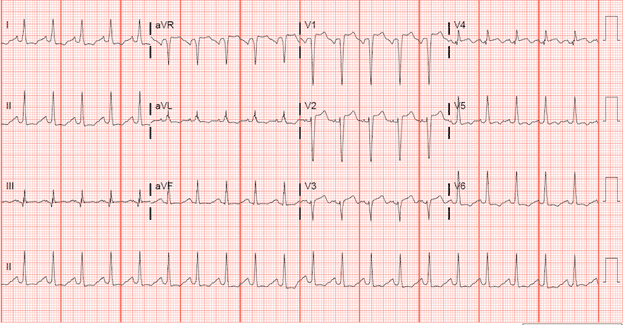
Initial Troponin 0.94
Left Ventricular Hypertrophy (LVH) review:The left ventricle hypertrophies in response increased pressure needed to overcome resistance from conditions such as hypertension, aortic stenosis, valvular abnormalities, or obstructive pathologies (HOCM). Numerous Criteria for diagnosing LVH: Sokolow-Lyon, Cornell, Modified Cornell, and Romhilt-Estes. Sokolow-Lyon is the most commonly used criteria and will be the one discussed below.
Criteria:
Voltage Criteria:
- S wave of V1 + tallest R wave between V5 and 6 >35 mm
- Limb Leads: R in I + S in lead III > 25 mm, R in aVL >11 mm, R in aVF >20 mm, S in aVR >14 mm
- Precordial Leads: R wave in V4,5, or 6 is >26 mm, R in V5 or V6 + S wave in V1 >35mm, or finally the largest R plus the largest precordial S is >45 mm
Non-Voltage Criteria
- Increased R wave peak time (“The time from the onset of the earliest Q or R `wave to the peak of the R wave”) > 50 ms in leads V5 or V6
- ST depression and T wave inversion in lateral leads
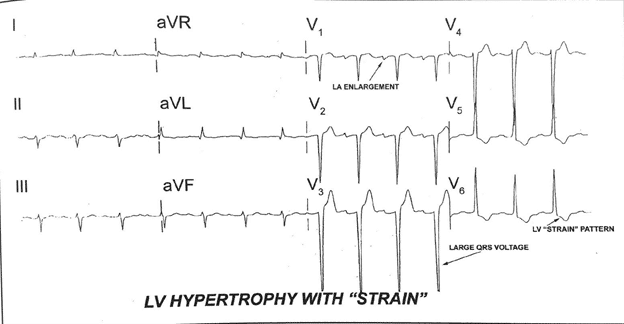
Another example from LITFL:
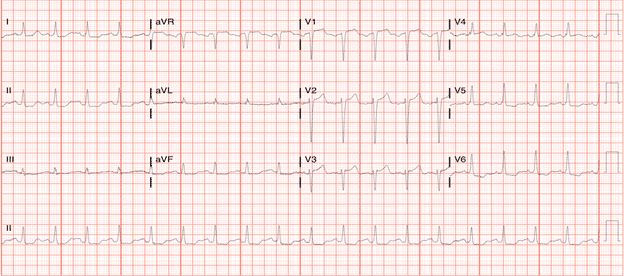
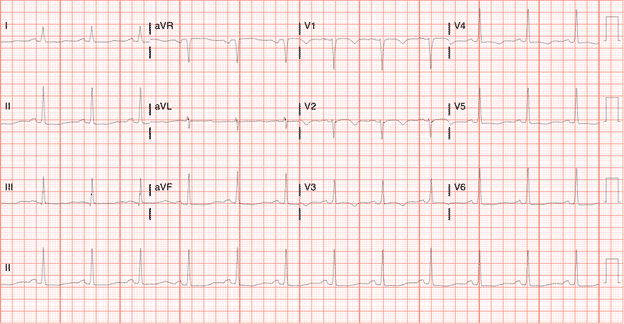
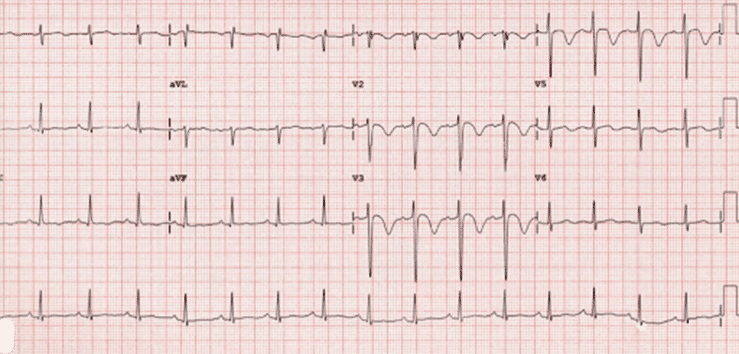
Patient’s repeat EKG
ICU course:
Pt was admitted to the ICU and placed in negative pressure isolation and was persistently hypotensive and hypoxic requiring FIO2 approximately 70-80%.
At the time, it was concluded that the EKG did not represent a STEMI. Her EKG met criteria for LVH and while V4 showed ST segment elevation it was not contiguous and the inferior ST segments were consistent with strain pattern. While the morphology was concerning, the patient’s persistent hypoxia with high FIO2 requirement and need for pressors to maintain blood pressure were likely the cause of the EKG changes and minimal troponin elevation.
Emdocs.net offered a potential pathway to assess for STEMI in the setting of LVH. This pathway was first described by Armstrong et al. http://www.emdocs.net/ecg-pointers-lvh-vs-stemi/lvh-vs-stemi/
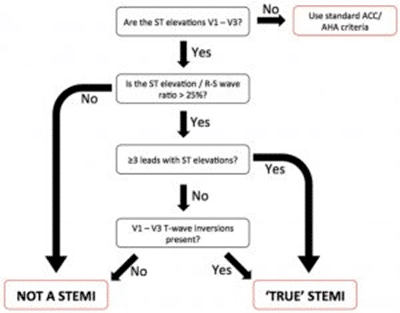
So when we apply this formula to the patient’s EKG:
The ST Elevation/R-S wave height is <25 % and, according to the study, not a true STEMI.
Cardiology was contacted who agreed with initial assessment that the patient did not meet STEMI criteria, and added that if the patient was experiencing ischemia/infarct they would expect the troponin elevation to be significantly higher than 9 and initial EKG changes/troponin elevation to likely be due to hypotension and hypoxia. Pt was given ASA, Plavix and heparin, and Cardiology would re-evaluate.
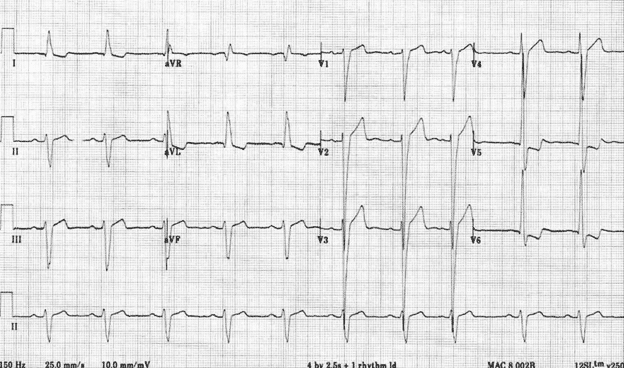
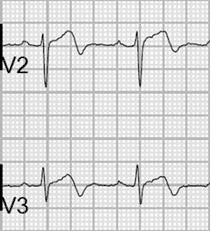
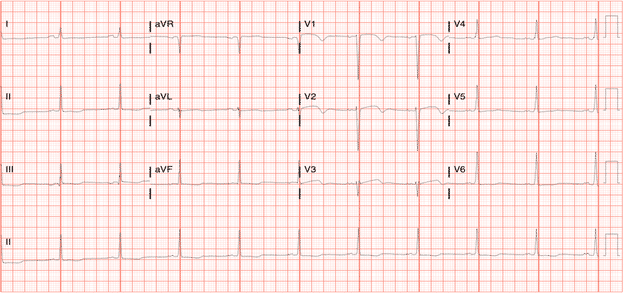
Noted on the 3rd EKG was a significant shift in morphology to include a Wellenoid appearing ST segment change.
EKG 3
The patients second troponin was approximately 9.
Troponin later rose to about 25 before declining substantially to 9 again the next day.
Wellen’s Review:
Wellens Syndrome is an EKG pattern highly specific and concerning for critical stenosis of the left anterior descending artery. The pattern most commonly involves biphasic or deeply inverted T waves in leads V2 and V3. Wellens syndrome often occurs when the patient is not experiencing chest pain, and the pattern can develop over days to weeks. While there have been multiple criteria proposed for diagnosis of Wellens, the most weight is placed on T wave morphology in leads V2 and V3. With biphasic T waves with terminal negative inflection, or deeply inverted T waves in V2 and V3, Wellens syndrome must be a major concern. Other diagnostic criteria that often present with this T wave morphology in Wellens include:
- Isoelectric or minimally-elevated ST segment (< 1mm)
- No precordial Q waves
- Preserved precordial R wave progression
- Recent history of angina
- ECG pattern present in pain-free state
- Normal or slightly elevated serum cardiac markers
There are two types of Wellens patterns on EKG, Type A and Type B.
Wellens Type A
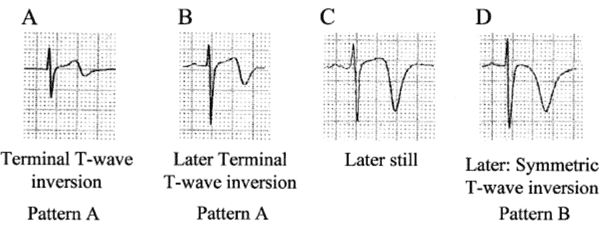
Type A Wellens pattern involves biphasic T waves with initial positivity and terminal negative deflection in lead V2 and V3. This pattern accounts for approximately 25% of Wellens Syndrome cases. Patients are often asymptomatic and may have little to no troponin elevation.
Wellens Type B
Type B Wellens pattern is characterized by deeply inverted T waves in lead V2 and V3. This pattern accounts for 75% of disease presentation. Patients are often still asymptomatic and have minimal troponin elevation on initial presentation. Wellens morphology exists on a spectrum, with EKG patterns progressing from Type A to Type B.
Treatment for Wellens is immediate PCI. As wellens is a preinfarction pattern resulting from critical LAD stenosis, failure to obtain urgent PCI could result in massive anterior wall myocardial infarction with poor prognosis. While awaiting cardiology consult and catheterization, is is recommended that Wellens be treated medically as an acute MI with antiplatelet therapy and heparin.
EKG 4
Continued ICU course
After her COVID tests resulted negative the patient was taken off of isolation precautions. Her bacterial sepsis therapy continued and her chest xray showed improvement in her pneumonia.
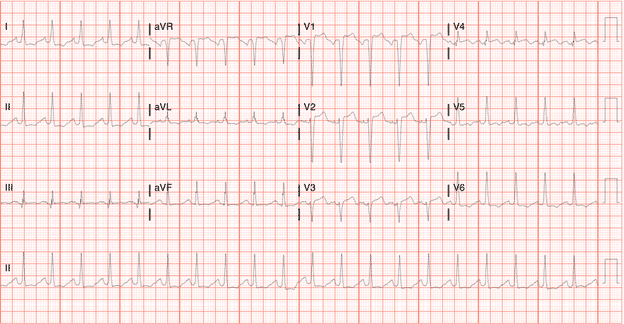
Later her nurse noted a morphology change on monitor and did an EKG to follow up.
The patient’s EKG had a reversion of her initial EKG pattern, which was suspicious as her blood pressure was no longer requiring vasopressor support and her FIO2 requirements had improved.
EKG 5

Troponin again started trending up and patient was taken for left heart catheterization and was found to have an occluded LAD with severe stenosis of the LCX and occluded RCA with good collateral flow. Stents were placed in the LAD x3 and one stent in the LCX.
Remainder of ICU course:
She continued to improve but suffered intermittent setbacks to include atrial fibrillation with RVR and pseudomonas sp. pulmonary infection. Ultimately, she was extubated and was discharged after a 20 day stay in the hospital and is doing well.
References
Armstrong, Ehrin J., et al. “Electrocardiographic Criteria for ST-Elevation Myocardial Infarction in Patients With Left Ventricular Hypertrophy.” The American Journal of Cardiology, vol. 110, no. 7, 2012, pp. 977–983., doi:10.1016/j.amjcard.2012.05.032.
Burns Emergency, Ed. “Left Ventricular Hypertrophy (LVH) • LITFL • ECG Library Diagnosis.” Life in the Fast Lane • LITFL • Medical Blog, 16 Mar. 2019, litfl.com/left-ventricular-hypertrophy-lvh-ecg-library/.
Cadogan, D., 2020. Wellens Syndrome • LITFL Medical Blog • ECG Library Eponym. [online] Life in the Fast Lane • LITFL • Medical Blog. Available at: <https://litfl.com/wellens-syndrome-ecg-library/> [Accessed 30 June 2020].
“ECG Pointers: LVH vs STEMI.” EmDOCs.net – Emergency Medicine Education, 15 Aug. 2018, www.emdocs.net/ecg-pointers-lvh-vs-stemi/.
Ramires TG, Sant’Anna J, Pais J, Picarra BC. Wellens’ syndrome: a pattern to remember. BMJ Case Rep. 2018 Apr 10;2018 [PMC free article] [PubMed
Rhinehardt J, Brady WJ, Perron AD, Mattu A. Electrocardiographic manifestations of Wellens syndrome. Am J Emerg Med. 2002 Nov;20(7):638-43. PubMed PMID: 12442245
Rau, Ramesh R. Clinical Cardiology Made Easy. Jaypee Brothers Medical Publishers (P) Limited, 2015. Figure 1 (figure 3-38 in text)
Singh D, Suliman I, Chyshkevych I, Dabage N. A Pathognomonic Electrocardiogram That Requires Urgent Percutaneous Intervention: A Case of Wellens Syndrome in a Previously Healthy 55-Year-Old Male. Am J Case Rep. 2019 Jan 28;20:117-120.