Background:
The overwhelming majority of cerebrovascular accidents, or strokes, are caused by underlying hypertension, hyperlipidemia, tobacco use, obesity, and diabetes (2). Stroke is a leading cause of death for Americans, and most strokes occur in people age 65 and older, however this age continues to grow younger with time, meaning clinicians have to continue to keep strokes on the differential for young patients and be vigilant in recognizing when it may be occurring in someone who doesn’t fit the typical demographic (3). Spontaneous intracerebral hemorrhage leading to stroke is a rare complication of pregnancy, occurring in only about 0.01% of all pregnancies, but when it occurs, it can be particularly devastating, as these patients are usually young and in good health (1). This report highlights the devastating aftermath of a peripartum hemorrhagic stroke in a young, otherwise healthy patient.
Case Presentation:
Patient is a 39 year old G6P6 Hispanic female with no past medical history, 7 days post-partum from an uncomplicated, full-term vaginal delivery, who presents to the ED for altered mental status. Patient was in her usual health when she decided to take a nap at 11PM. When family members attempted to wake her at 3:00 AM the following morning, she was minimally responsive, not answering questions, and not visualized to be moving any of her extremities, although she was spontaneously breathing and had a pulse. Per discussion with family, the patient had started to complain of headaches two days postpartum and was admitted at an outside hospital for pre-eclampsia, where she was treated with magnesium, observed for 72 hours, and then discharged home, per report, without any antihypertensive medications. Per husband, patient’s blood pressures remained high over the remaining couple of days
post-discharge, with reports of systolics in the 220s.
Vitals/Physical exam:
BP 199/103 (MAP 143). HR 63. RR 20. T 98.7℉. Young Hispanic female, awake but not alert, not making any purposeful movements. Patient is disoriented and ill-appearing. Unable to answer questions. Normocephalic, atraumatic. No evidence of external head trauma. Pupils are 4 mm and round bilaterally, however both are unreactive. No nystagmus. Patient opens eyes spontaneously but does not track. She has a regular rate and rhythm. Breath sounds are clear
bilaterally. She is not seen moving any of her extremities spontaneously or to painful stimuli. Right upper extremity appears flexed and contractured. Hypertonic upper and lower extremities bilaterally. Left foot with clonus. She has a right-sided facial droop. GCS of 6 (E4V1M1).
Labs/Imaging:
Patient was immediately activated as a Code Stroke, and therefore imaging was obtained prior to labs.
Imaging is shown below.

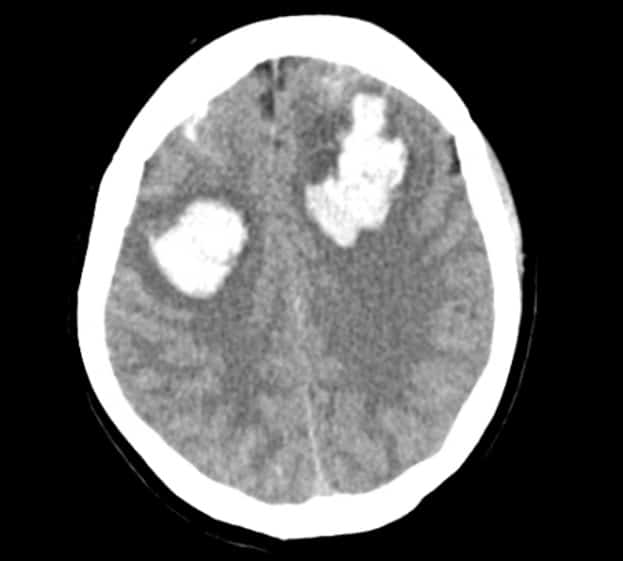
CT Head without contrast – showing large bilateral frontal lobe intraparenchymal acute hyperdense hemorrhages, along with subarachnoid blood in the bilateral frontal lobes and right parietal lobe. There is also a small subdural hematoma along the medial superior bilateral frontal lobes. Additionally, there is a 4mm rightward midline shift.
Initial labs were largely unremarkable. Labs notable for: Slight leukocytosis of 12.8. Hgb 13.1. Platelets of 266. K of 3.9 initially. Na of 138. Calcium of 8.8. Slightly hyperglycemic at 131. BUN of 12 and Cr of 0.60. PT 11.8, PTT 29.9 and INR of 1.04. Phosphorus normal at 3.4. Magnesium level was elevated at 4.2. Fibrinogen slightly elevated at 414, with slight elevation in LDH as well at 268. Haptoglobin normal at 189. Total bilirubin of 0.3, with direct bilirubin 0.07 and indirect bilirubin 0.23. Alkaline phosphatase 104, AST 28 and ALT 34. Patient was incidentally found to be COVID-19 positive.
Clinical Course:
Patient was intubated in the emergency room shortly after arrival. Neurosurgery was consulted emergently and recommended obtaining CTA and CTV scans, which were ultimately negative for aneurysm, vascular malformation, or venous sinus thrombosis. A cardene drip was initiated to maintain systolic blood pressures below 160. Obstetrics and Gynecology was also consulted, and they recommended beginning magnesium for seizure prophylaxis and for presumed hemorrhagic stroke secondary to eclampsia. Patient was admitted to the Medical ICU for close neurological observation and management of ventilation.
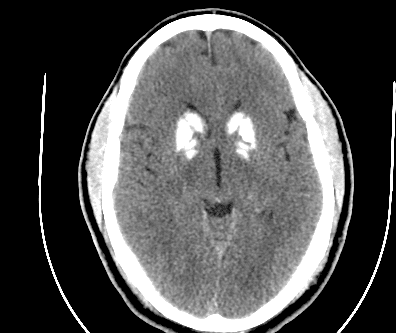
Repeat CT head scan six hour post-initial scan showed similar size of the bifrontal hematomas, subarachnoid, and subdural bleeding, with early intraventricular decompression of the left frontal hematoma into the left lateral ventricle.
On HD#1, per Neurosurgery’s recommendations, an external ventricular drain (EVD) was placed for continuous intracranial pressure monitoring and CSF drainage as needed. Initial supine ICP read as 26 mmHg (normal is 7-15 mmHg, with values >15 considered abnormal and values >20 considered pathological [4]). Orders placed to leave EVD open to drain at +15 cmH20 and to continue hyperosmolar therapy and sedation as needed to maintain goal ICP < 20 and goal CPP of 70 mmHg. Magnesium IV was given for a total of 24 hours and then discontinued per protocol and patient was then switched to empiric levetiracetam.
On HD#2, there was concern for peri-herniation, with recurrent bradycardia and hypertension. Additionally, patient became febrile to 100.6F. A 23.5% hypertonic saline bullet was administered. Cardene was continued until HD#3, when patient was titrated off and transitioned to oral labetalol.
Additionally, the patient began to develop seizure-like activity (posturing) on HD#3. EEG showed diffuse background slowing, likely due to toxic metabolic degenerative hypoxia, with no clear epileptiform activity. These symptoms were thought to be due to agitation, as ICP curves showed significant jumps during these episodes. Patient was initially well-sedated on propofol but required unclamping of the EVD due to increased ICPs (into 30s) and addition of fentanyl drip.
Patient was not tolerating clamping trials and ICPs remained elevated on HD#4, and patient was given a one-time dose of 20% mannitol and continuous 3% sodium chloride. Meanwhile, a cooling catheter was placed for fevers and a full sepsis workup was ordered. Patient discovered to have MRSA pneumonia and placed on broad-spectrum.
On HD#6, patient found to have sustained ICPs in the 30s despite increased sedation and hypertonic interventions. There was no associated hydrocephalus as a cause for this spike. At the same time, patient was very agitated and dyssynchronous with the vent, requiring additional sedation with versed and precedex, in addition to her fentanyl and propofol. There was concern that the patient had developed a neurogenic storm. Patient was ultimately placed into a pentobarbital coma, which was successful in reducing ICPs, however greatly decreased patient’s mean arterial pressures, resulting in patient being placed on norepinephrine for vasopressor support.
Pentobarbital was discontinued HD#7. Additionally, patient developed severe edema and decreased urine output. Leukocytosis worsened to 23K. There was low concern for bolt infection by Neurosurgery. Blood cultures and CSF cultures showed no growth. She was found to have bilateral pulmonary opacities on CT imaging, with increase in O2 requirements on the ventilator. This was thought to be due to pulmonary edema versus cardiogenic edema versus neurogenic edema versus COVID-19 pneumonia with acute respiratory distress syndrome (ARDS). She was started on furosemide IV, remdesivir, and dexamethasone.
By HD#12, patient had developed a severe thrombocytopenia, anemia, oliguria, worsening respiratory status on the ventilator, and concern for neurogenic storm. At the time of case report submission, patient remained Full Code in the ICU as she waited for family members to arrive from Mexico to discuss goals of care.
Discussion and Review of Eclampsia:
Pre-eclampsia and eclampsia are both complications seen in late pregnancy, caused by pregnancy-induced hypertension. Approximately 2-7% of pregnancies are complicated by pregnancy-induced hypertension. Gestational hypertension is defined as a new blood pressure reading of 140/90 or higher, which resolves in the postpartum period. Pre-eclampsia is gestational hypertension with proteinuria (>300mg/24hr), while eclampsia results when seizures occur in a patient with signs of pre-eclampsia (5). In cases of severe eclampsia, patients may complain of headaches and visual disturbances and they can develop diastolic blood pressures over 110 mmHg, severe proteinuria, thrombocytopenia, transaminitis, and oliguria. The most dangerous complication of eclampsia is the development of seizures. In postpartum women who have eclampsia, 55% have not been previously diagnosed with pre-eclampsia. The progression from pre-eclampsia to eclampsia is often quick and unpredictable and is a major cause of maternal mortality. Patients with severe post-partum pre-eclampsia or in whom you suspect eclampsia, the patient should have 2 peripheral IVs placed upon arrival. Blood tests should include CBC, renal function studies, liver function tests, platelet counts, a baseline magnesium level, and a coagulation profile. Aside from devastating intracranial hemorrhage, another complication of preeclampsia is the development of HELLP Syndrome, which is characterized by hemolysis, elevated liver enzymes (AST and ALT > 70), and low platelet count (<100,000). Management of eclampsia in the pre-delivery stage is delivery of the fetus. However, in the case of the patient involved in the case, who was postpartum, the goal is to control seizures and control hypertension to prevent permanent damage to maternal organs, and an outcome such as an intracranial hemorrhage. As with all seizure patients, you must exclude other likely causes of seizures, such as hypoglycemia and drug abuse. Seizures associated with eclampsia are almost always controlled by magnesium sulfate IV. It has very little antihypertensive effect but is the most effective anticonvulsant and often the hypertension associated with eclampsia resolves with control of seizures. An IV loading dose of 4g magnesium, followed by 2g/hr IV for a total of 24 hours, is recommended. Hypermagnesemia can lead to loss of reflexes or respiratory depression, which can be reversed with 1g slow IV push of calcium gluconate. The antihypertensives used primarily in a pre-eclamptic or eclamptic patient, especially during pregnancy, include hydralazine, labetalol and nifedipine. Of course, in a patient who presents with intracranial hemorrhage as in the case described, management is centered around conventional interventions for ICH. Advanced neuroimaging with CTA and CTV is important to rule out secondary causes, such as aneurysm, AVM, fistula, or venous sinus thrombosis. Blood pressure control is paramount as hypertension can worsen bleeding, however it’s important not to lower the blood pressure too much thereby reducing cerebral blood flow. The current consensus regarding blood pressure is to provide antihypertensive treatment with parenteral agents for systolic blood pressures higher than 160 mmHg and MAP higher than 130 mmHg.
Neurosurgery should be consulted. For patients with clinical or radiographic evidence of elevated intracranial pressure, therapies to lower ICP should be considered, including EVD placement or hematoma evacuation. Seizure activity can worsen neuronal injury and elevations in ICP, therefore patients with known or suspected seizures should be placed on antiepileptic drugs. And finally, as with any other type of stroke, general supportive care should be provided to minimize neuronal injury, which includes treating any hyperthermia or signs of neurogenic storm, which often presents as increased posturing, agitation, hypertension, hyperthermia, tachycardia, and tachypnea (6).
Conclusion:
This summarizes a report of a young, otherwise healthy female, one-week status post an uncomplicated vaginal delivery, who then developed headaches and elevated blood pressures and ultimately presented to the emergency department with altered mental status and was found to have suffered a hemorrhagic stroke secondary to eclampsia. Patient did develop seizures shortly after admission. Patient completed a course of IV magnesium sulfate for seizure management related to postpartum eclampsia and was then placed on AEDs. Additionally, patient initially required further blood pressure control for her hemorrhagic stroke and was managed with nicardipine (as opposed to labetalol, hydralazine, and nifedipine, which would be used in the pregnant patient). An EVD was placed and patient’s ICU course was complicated by increased intracranial pressures, which required further sedatives and a brief pentobarbital coma, which then resulted in hypotension requiring pressor support. She additionally developed fevers, MRSA pneumonia, pulmonary edema thought to be possibly related to her incidental COVID+ status, as well as neurogenic storm. Patient’s prognosis at the time of the report was extremely poor, and palliative care physicians were waiting for family
members to arrive internationally to discuss goals of care.
References:
- https://www.ahajournals.org/doi/full/10.1161/STROKEAHA.118.023118
- https://www.cdc.gov/stroke/facts.htm
- file:///Users/rachelantol/Downloads/stroke-young-patients-qa.pdf
- https://emedicine.medscape.com/article/1829950-overview
- https://expertconsult.inkling.com/read/wallsrosens-emergency-medicine-concepts-clinical
-practice-9/chapter-178/complications-of-late-pregnancy - https://pubmed.ncbi.nlm.nih.gov/14998101/


This Post Has 0 Comments